In the world of clinical trials, achieving real-time end-to-end visibility has become more than just a trend; it’s a critical necessity.
Continue readingIn the world of clinical trials, achieving real-time end-to-end visibility has become more than just a trend; it’s a critical necessity.
Continue reading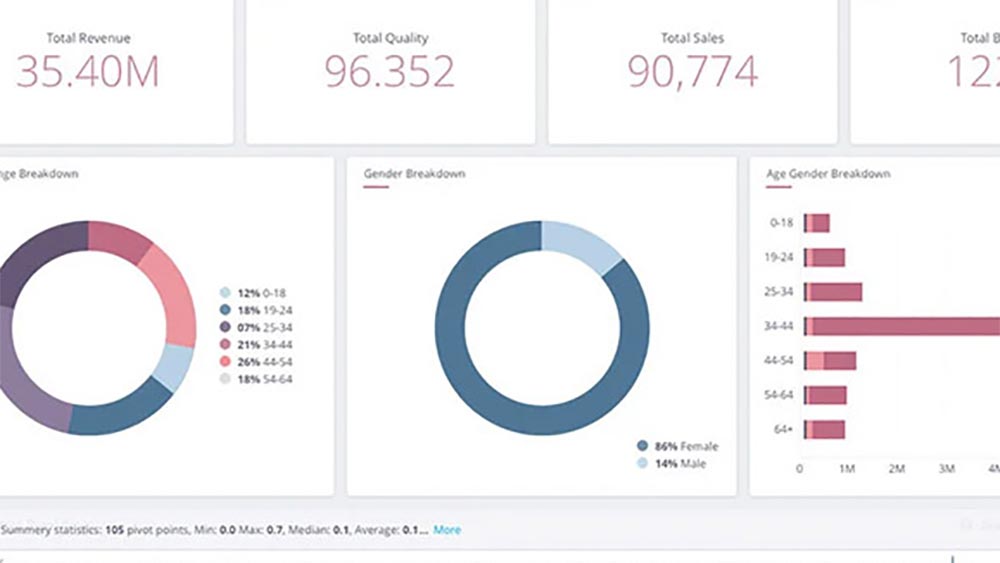
Error: Contact form not found.
Error: Contact form not found.
Error: Contact form not found.
Error: Contact form not found.
[email-download download_id=”9583″ contact_form_id=”7713″]
[email-download download_id=”9446″ contact_form_id=”7713″]
Error: Contact form not found.
Error: Contact form not found.
Error: Contact form not found.
The retailer client wanted a data-driven scientific approach to unlock business potential. This platform would take into account multiple predefined business dimensions to enable its reporting and business KPI dashboard and unlock answers for critical business questions via deployment of analytics solutions on a single platform tcg mcube. With Artificial Intelligence and Machine Learning capabilities built in the platform, this project would lay the foundation of a powerful data science journey and create differentiated value.The retailer client wanted a data-driven scientific approach to unlock business potential. This platform would take into account multiple predefined business dimensions to enable its reporting and business KPI dashboard and unlock answers for critical business questions via deployment of analytics solutions on a single platform tcg mcube. With Artificial Intelligence and Machine Learning capabilities built in the platform, this project would lay the foundation of a powerful data science journey and create differentiated value.
