Large language models (LLMs) stand to redefine enterprise AI with GPT, Bard, Gemini, and Llama 2 generating human-like text and catering to knowledge management applications.
Continue readingRevolutionizing Flow Cytometry with AI and Machine Learning
Flow cytometry is a formidable asset for a researcher. By compartmentalizing cells based on set molecular characteristics, flow cytometry provides information on specific cell types from highly complex and populated samples. This technique analyzes thousands of cells per second, allowing researchers to collect huge data volumes in a relatively short time span.
Continue readingModernize your legacy mainframe systems
Many organizations use legacy mainframe in their datacentres to run critical operations. This age-old system tuned and customized to the functional requirements, locked-in to vendors over the years, has been used extensively across business critical operations.
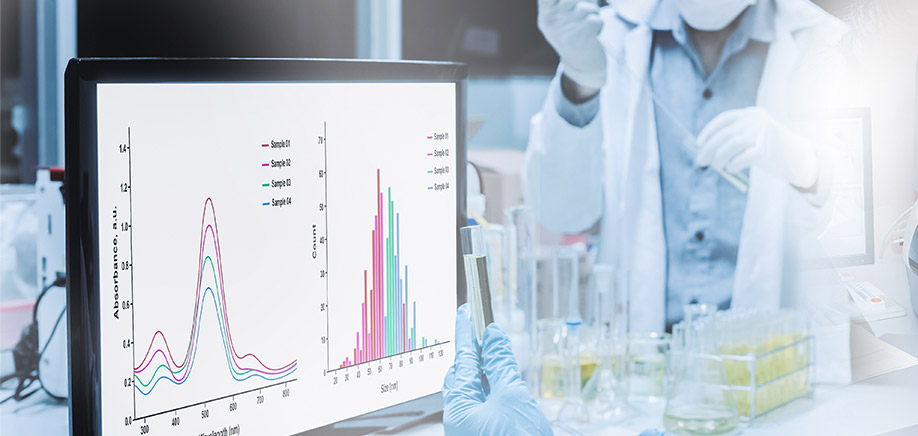
Continue readingFrom Bench to Breakthrough: Revolutionizing Drug Research with AI-Powered Integrated Assay Analysis
In the dynamic landscape of life sciences research, the acceleration of discoveries and breakthroughs is of paramount importance. Today, integrated assay analysis, powered by cutting-edge AI solutions, represents a pivotal leap in scientific exploration, revolutionizing the pace, depth, and efficacy of research endeavors across diverse disciplines.
Continue reading